
One of the most common compounds produced by elemental hydrogen is hydrogen peroxide (H 2O 2) with oxygen. It can lead to the formation of hydrides of various metals, non-metals such as, H 2S, NAH, PH 3 and KH. Free hydrogen radical can react with chlorides and oxides of various metals to form free metals. And lead to the dissociation of its diatomic form into free atom of hydrogen, which is highly reactive and a powerful reducing agent. Hydrogen becomes highly reactive at higher temperatures. However, under different environmental conditions, various metals absorb hydrogen, such as the absorption of hydrogen by steel can cause brittleness in steel. At room temperature, hydrogen is stable and unreactive. Hydrogen is more soluble in organic solvents and less soluble in water. Hydrogen is a highly (combustible) flammable gas. It has a boiling point of -252.8C and a melting point of -259.2 C. Hydrogen has an atomic number of 1 and atomic mass of 1.007825g.mol -1. It is non-toxic and nonmetallic at standard conditions (standard temperature and pressure). It has the lowest density (0.0899*10 -3 g.cm -3 at 20 ☌) as compared to all other gases. Hydrogen gas is the lightest of all gases. The neutral atomic state of hydrogen is mainly present in the interstellar medium ISM (outer space). In its plasma form, hydrogen give rise to natural phenomenon of Birkeland currents and aurora (by interacting with magnetosphere of the Earth). Hydrogen is mostly present in its atomic and plasma states, which have significantly different properties as compared to the molecular (gaseous) form. 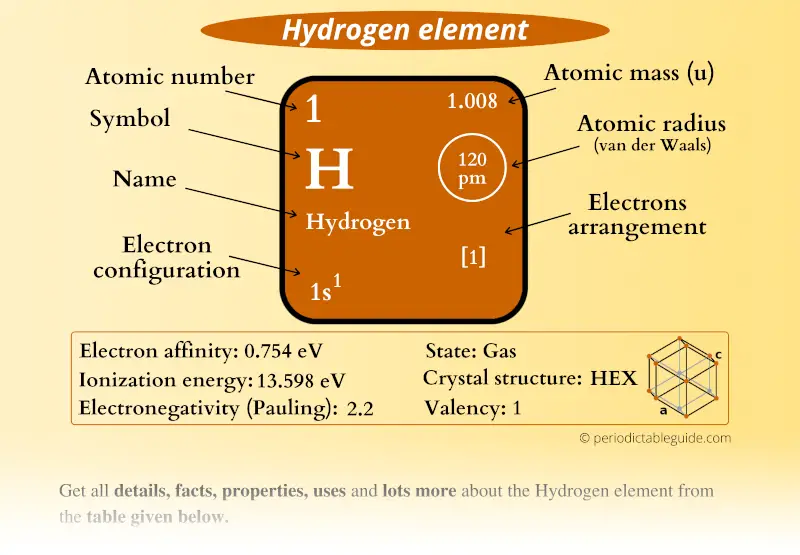
In gaseous form, it is present only in a small fraction and makes around 1 part per million in volume. Hydrogen is found in significant abundance in undetected form of mass, such as dark matter, and in gas giant planets and stars. Hydrogen is the main component of living systems (hydrocarbons). It is considered as the 3 rd most abundant element in the Earth’s atmosphere and the most abundant element in the universe. Hydrogen is ubiquitous in nature as it is present in water. Hydrogen also has a notorious event related to its name, the incident of Hindenburg airship (1937), were combustion of hydrogen led to the destruction of the ship in the midair and brought an end to the era of hydrogen-based air-travels. Hydrogen-lifted airships found tremendous usage in the World War 1, where they were used for transferring people, as observation platforms and as bombers. And later, Henri Giffard (1852), made the first hydrogen-lifted airship. In as early as 1783, hydrogen gas was used in air balloons by Jacques Charles, which demonstrated the power and reliability of hydrogen for providing the lift for air-travel. In 1898, James Dewar successfully liquified hydrogen. Hence the gas was named hydro-genes (1783 by Antoine Lavoisier), which is the Greek word for “water-former”. But the formal discovery of hydrogen was reported by Henry Cavendish (1766), who analyzed the bubbles and showed that burning of hydrogen lead to the production of water. 
The informal discovery of hydrogen dates to 1500s when Paracelsus (alchemist) observed the production of bubbles (gas) when sulfuric acid was added to iron and later Robert Boyle (1671) observed the production of same flammable bubbles. It was discovered in 1766 by Henry Cavendish and is widely used for various industrial, medical and recreational purposes. Hydrogen is one of the three most abundant elements present on Earth.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
